Technology
‘MedTech Mitra’ Launched: This Initiative Will Help Medical Innovators Translate Ideas To Products For Patients In Need
Karan Kamble
Dec 25, 2023, 07:50 PM | Updated 07:53 PM IST
Save & read from anywhere!
Bookmark stories for easy access on any device or the Swarajya app.


Medical technology, or medtech, innovators can now expect some much-needed hand-holding from the government through the often long and arduous process from idea to market-ready product for patients in need.
The Indian government on Monday (25 December) launched “MedTech Mitra” — a novel platform to provide crucial assistance to the country’s medtech innovators through the various stages of clinical evaluation, regulatory facilitation, and uptake of new products.
The key players involved in the initiative are the Indian Council of Medical Research (ICMR), the Central Drugs Standard Control Organisation (CDSCO), and the government think tank NITI Aayog.
“ICMR and CDSCO are fully committed to taking the innovations of Indian medical innovators to the people of India for their benefit, for their health. That is our mission,” Dr Rajiv Bahl, Secretary to the Department of Health Research and Director General of ICMR, said at the launch.
“This is our one contribution to Prime Minister Narendra Modi’s call for Aatmanirbhar Bharat and Viksit Bharat,” he added.
Why “MedTech Mitra”
For a medical innovator, the road from a novel idea to a medical product, such as a device, can be very bumpy, threatening to derail the entire journey at every step.
This situation is concerning because of the tremendous value of excellent, homegrown medical solutions — say, a new vaccine or a cancer cure — that can be made available to the 140 crore people in India who, above all, want to lead healthy lives.
Great medical ideas take birth in scientific laboratories, academic institutions, and startups, but, upon facing various challenges along the way, get stuck in roadblocks and end up going nowhere helpful.

“The cures don’t reach the patient. They keep circling around in this long process. A lot of time also passes. So, it’s a loss. The country’s capability to provide good health is not fulfilled,” said Dr Vinod K Paul, a Member of NITI Aayog.
Recognising this critical issue, government agencies have come up with MedTech Mitra, a platform to help medical innovators go around various roadblocks and swiftly reach their destination — getting the medical device out there for patients in need.
“We have to build a bridge over this valley of death so that whatever possibilities emerge from innovators reach the patients as soon as possible,” Paul said, referencing the 2008 paper ‘Translational research: Crossing the valley of death’.
The valley of death represents the huge gulf between the possible cures available in scientists’ hands and the patients who need them in hospitals.
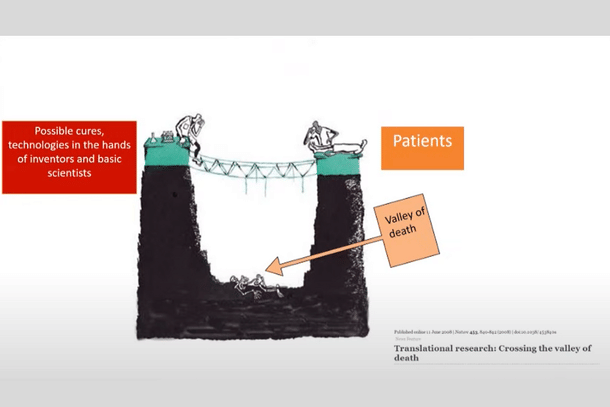
“An innovator who has good intentions to serve their country and possesses passion, strength, and skill has to face many difficulties in their path, and these difficulties stall progress. Our MedTech Mitra system is to help complete this journey,” Paul explained.
MedTech Mitra can help out in the major stages like regulatory approval, preclinical and clinical trials, including animal trials, aligning the product to existing guidelines, and the assessment of health technology and the product’s cost-effectiveness.
Notably, the hand-holding progress can begin at any stage of the life cycle — from idea to proof of concept to prototype to product to public access. Thus, an innovator can seek help at any stage — even right at the beginning of the process.
“The biggest work that DHR-ICMR will do, which no one else will be able to do, is to conduct clinical trials,” Paul said.
As for the innovators seeking help, they have to get on to the MedTech Mitra portal to fill up an online form and make a submission.
A group from the government’s side will check the form, initiate discussions, and even carry out in-person meetings if the need arises.
They will then determine the use case for the technology, such as in primary care or as an assistive technology. Financial support mechanisms will also be explored, altogether forming an end-to-end guidance process.

India’s Medtech Sector
India’s medtech sector faces challenges in converting innovations to products due to regulatory issues, compounded by a lack of collaboration between engineers and clinicians.
Thus, while moving the dial towards universal health coverage, the MedTech Mitra initiative will provide much-needed impetus to the development of this key sector, recognised as a sunrise sector under the Make in India initiative.
India happens to be a fast-growing medical devices market — the fastest among the emerging markets — with a 1.5 share in the global market. A greater focus on research and development, specifically innovation, can help India lift its market share to 4 per cent.
This growth will be crucial for India since, for medical devices, it has a 75-80 per cent import dependency. “This collaborative initiative will facilitate indigenous development of affordable, quality medtech devices and diagnostics, leading to a considerable reduction in the import dependence of this sector,” Dr Mansukh Mandaviya, the Minister of Health and Family Welfare, said at the launch, which coincided with Good Governance Day.
“MedTech Mitra is a platform for budding entrepreneurs and innovators in India. It is more than an ecosystem, a community. It is a harbinger of revolutionary change,” said Professor S P Singh Bhagel, the Minister of State for Health and Family Welfare, who was also present at the launch.
“MedTech Mitra will empower emerging startups and ensure ease of innovation, ease of doing research and development, ease of rendering service in building an Aatmanirbhar Bharat,” said Paul, adding that “in harbouring collaboration among all the stakeholders, it will effectively break silos, catalysing growth and independence in this sector.”
But it will all begin by taking many more medical ideas successfully from ‘the lab bench to the hospital bedside’.
MedTech Mitra is, thus, a significant step in promoting indigenous development of health technology, and in line with Prime Minister Narendra Modi’s vision of an aatmanirbhar Bharat (self-reliant India).
Karan Kamble writes on science and technology. He occasionally wears the hat of a video anchor for Swarajya's online video programmes.




