Insta
Indian Covaxin To Boost Mexico's Fight Against Covid-19, Gets Emergency Use Approval From Mexican Drug Regulator
Swarajya Staff
Apr 07, 2021, 11:41 AM | Updated 11:41 AM IST
Save & read from anywhere!
Bookmark stories for easy access on any device or the Swarajya app.

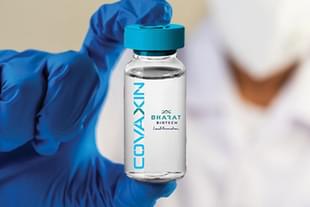
In a big boost for India's first indigenous Covaxin Covid-19 vaccine, the drug regulator of Mexico has approved the emergency use of the vaccine in the Latin American country.
The Covaxin has been developed by Hyderabad-based Bharat Biotech in collaboration with the Indian Council of Medical Research (ICMR), and is one of the two vaccines currently being employed in India's Covid-19 vaccination drive. The other Covid-19 vaccine that is being used in India is Serum Institute of India's Covishield.
The Mexican drug regulator, COFEPRIS, has authorized the emergency use of Covaxin, Mexican Foreign Minister Marcelo Ebrard said on Tuesday (7 April).
"Very timely decision by COFEPRIS to authorize the emergency use of COVAXINE vaccine manufactured in India. The options for vaccination against COVID-19 in Mexico are expaning !!!" Ebrard tweeted.
Bharat Biotech's Covaxin is the sixth Covid-19 vaccine that has been greenlighted for emergency use in Mexico. The other firms that got approval for their Covid-19 vaccines in Mexico are AstraZeneca, Cansino, Pfizer, Sinovac and Sputnik V.




