Ideas
A Tale Of Three Potential Vaccines – How They Stack Up In The Race For Coronavirus Prevention
Karan Kamble
Jul 24, 2020, 12:49 PM | Updated Jul 27, 2020, 09:00 AM IST
Save & read from anywhere!
Bookmark stories for easy access on any device or the Swarajya app.
The genetic sequences for the novel coronavirus were made public a little over six months ago. Since then, vaccine manufacturers around the world have been sprinting hard and fast to arrive at a drug that can stop the coronavirus SARS-CoV-2 in its tracks.
Currently, there are 142 candidate vaccines (as on 21 July) in the pre-clinical evaluation stage – which means that they are being tested in a lab or non-human animals – and 24 in clinical evaluation.
A handful of vaccines among these probables has emerged as the most promising – or generating the most hype if you will. Narrowing the stream down further, three candidates, in particular, are at the moment making the right noises.
- The Oxford University vaccine in partnership with UK pharmaceutical major AstraZeneca
- The vaccine by pharmaceutical major Pfizer and European biotech company BioNTech
- US company Moderna’s vaccine in collaboration with the National Institutes of Health
Vaccine Development
ChAdOx1 nCoV-19
The team at Oxford led by professors Sarah Gilbert, Andrew Pollard, Teresa Lambe, and others started work on a vaccine on 10 January, around the time that the World Health Organization (WHO) received the genetic information for the virus. A chimpanzee adenovirus vaccine vector (ChAdOx1), developed at the in-house Jenner Institute, was picked as a suitable technology for developing the SARS-CoV-2 vaccine.
This virus vector is well-studied and has been used across a wide range of ages, as Oxford reports, “from 1 week to 90 years of age, in vaccines targeting over 10 different diseases.”
This isn’t Professor Gilbert and the team’s first crack at this work as they have previously worked on a vaccine for another coronavirus, one which causes the Middle East Respiratory Syndrome.
Two and a half months of vaccine development ensued and the Oxford researchers began screening healthy volunteers in the age bracket of 18-55 years for a clinical trial.
In April, testing began for volunteers. The pool was 1,077 people, half of whom received the vaccine, and the other half (the control group) a meningitis vaccine.
As testing and analysis got underway, the University of Oxford announced its partnership with the UK-based global biopharmaceutical company AstraZeneca.
Then, on 20 July, it was reported that the Oxford coronavirus vaccine produced a strong immune response. It provoked a T cell response within 14 days of vaccination and an antibody response within 28 days. (Read how T cells work).
A strong immune response was observed in 10 volunteers who received two doses of the vaccine, hinting at the direction this vaccine would take in terms of the number of doses if it successfully navigated the various stages of testing.
Towards the end of May, Oxford had already made the announcement of phase 2/3 human trials. These phases will involve testing of a much larger group of people and, more importantly, across a wider age group, to include children and elderly people.
Trials have already begun in Brazil and South Africa. Testing of 30,000 patients is expected in the US as well. With the vaccine raising hope, the UK, the US, and others including Serum Institute of India (world’s largest vaccine producer by volume) have already entered into commitments with Oxford University and AstraZeneca to have more than 2 billion vaccine doses supplied to them.
BNT162
Pfizer announced its five-point plan to battle Covid-19 on 13 March. Chairman and chief executive officer (CEO) Dr Albert Bourla called for collaboration in the fight against the virus and announced that his company was up to the task – “Pfizer is working to advance our own potential antiviral therapies and is engaged with BioNTech on a potential mRNA coronavirus vaccine.”
Three days later, Pfizer and BioNTech made it official that the two companies would jointly develop BioNTech’s mRNA-based vaccine candidate BNT162. This collaboration was natural since both Pfizer and BioNTech had been involved in joint research and development work for a couple of years already to develop mRNA-based vaccines for prevention of influenza.
The BNT162 Covid-19 vaccine programme had multiple mRNA candidates. Progress was noted with antiviral activity recorded against SARS-CoV-2 in preclinical screening.
By 22 April, the phase 1/2 clinical trial for the vaccine – with its four mRNA candidates – received a nod from the German regulatory authority, the Paul-Ehrlich-Institut. The trial kicked off immediately after that in Germany and, after approvals, a week later in the US.
Preliminary data for a sub-group of 45 people arrived on 1 July. In an encouraging development, one of the four vaccine candidates, BNT162b1, was able to stimulate antibody responses at or above the levels observed in Covid-19-recovered plasma patients. Based on this data, the potential vaccines BNT162b1 and BNT162b2 were put on fast track by the US Food and Drug Administration.
Preliminary data from the phase 1/2 trial for 60 people in Germany backed up the US data, demonstrating the ability of BNT162b1 to elicit high SARS-CoV-2 neutralising titers.
In addition, T cell responses were observed for the first time for any Pfizer-BioNTech candidate.
On the back of the good results, both the UK and the US entered into agreements with Pfizer and BioNTech to procure an initial 30 million and 100 million doses of their vaccine respectively after the necessary approvals.
As more data trickles in from the phase 1/2 trials, the collaborators get to pick their lead vaccine candidate and decide on the dose level to be administered to a much larger group – possibly involving up to 30,000 volunteers – in succeeding trials, slated to get underway in July.
Efforts have been on to ramp up manufacturing capabilities to enhance the production capacity. The Pfizer-BioNTech combine is currently expecting to manufacture “up to 100 million doses by the end of 2020 and potentially more than 1.3 billion doses by the end of 2021”.
mRNA-1273
The American biotechnology company Moderna in collaboration with the National Institutes of Health finalised the sequence for their vaccine candidate mRNA-1273 two days after the genetic sequence of the novel coronavirus was made known by China.
In an astounding achievement, the company took just 25 days from sequence selection to vaccine manufacture. To draw a comparison, it took 20 months to develop a vaccine for the coronavirus that caused the Severe Acute Respiratory Syndrome (SARS) in 2002-03 – merely for testing in humans.
The pace of vaccine development was quick, but news of funding began to come in quickly too. For instance, in the third week of January, the Coalition for Epidemic Preparedness Innovations was roped in to fund the manufacture of Moderna’s vaccine.
Biomedical Advanced Research and Development Authority (BARDA) within the US Department of Health and Human Services came on board with a commitment of up to $483 million to push along the development of the potential vaccine.
Later, Moderna also struck a deal with Swiss company Lonza to enable manufacturing of up to one billion shots at the dose level of 50 µg per year.
The National Institute of Allergy and Infectious Diseases conducted the phase 1 trial in the US. On 24 February, the first batch of mRNA-1273 was released for testing. The first of the 45 patients were given a shot on 16 March. Simultaneously, work was underway to manufacture more doses for potential future trials.
On 12 May, Moderna received the ‘fast track’ designation from the US FDA for its potential vaccine. Positive preliminary results emerged from phase 1 almost a week later, with the mRNA-1273 eliciting neutralising antibody titer levels in the eight initial participants – four of whom received a 25 µg dose while the other four received a 100 µg dose.
The phase 2 study, with 600 healthy volunteers in two age groups of 18-55 years and 55 years and above, got underway towards the end of May.
On 14 July, the interim analysis of Moderna’s phase 1 study was published in The New England Journal of Medicine. The early results of the study showed rapid and strong immune responses against SARS-CoV-2 across all dose levels.
Moderna has already advanced the late-stage development of its Covid-19 vaccine. Its phase 3 study protocol has been made known too and, according to the company, reviewed by the US FDA.
“The randomized, 1:1 placebo-controlled trial is expected to include approximately 30,000 participants at the 100 µg dose level in the U.S,” revealed the company.
At 100 µg, Moderna expects to deliver around 500 million doses per year, and “possibly up to 1 billion doses per year, beginning in 2021”, according to the company.
Road Ahead
The title of a 14 July editorial in The New England Journal of Medicine, “The COVID-19 Vaccine-Development Multiverse”, captures faithfully this exceptionally diverse vaccine work in progress simultaneously across the world.
A vaccine usually takes 10 years to develop, according to the UK-based health research foundation Wellcome Trust.
In light of this fact, the speed in the case of Covid-19 vaccine is clearly unprecedented, indicating a departure from the old paradigm.
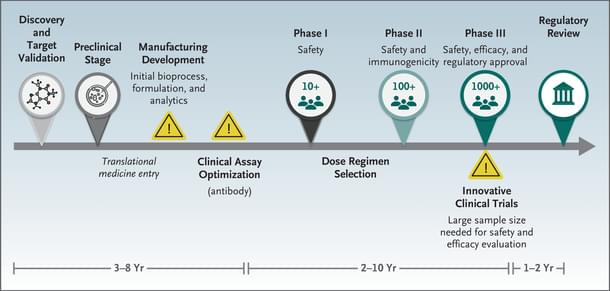
How optimistic should we be with respect to these vaccines, especially the three detailed in this story?
“The Oxford-AstraZeneca vaccine and the Moderna-NIH vaccine, based on their respective papers in The Lancet and The New England Journal of Medicine, make an excellent beginning,” says Dr Indranill Basu Ray, Professor of Public Health at the University of Memphis, Tennessee, US.
“However,” he adds, “there are certainly many things we do not know right now that will make us believe that these vaccines would make for a successful deterrent to COVID-19 in the actual practice scenario.”
As is the cliche, time will tell. Luckily for us, it probably won’t take long.
Karan Kamble writes on science and technology. He occasionally wears the hat of a video anchor for Swarajya's online video programmes.





